Corrosion Rate Calculation
What is Corrosion Rate?
Corrosion rate refers to the speed at which a material deteriorates due to chemical or electrochemical reactions with its environment. It qualifies material deterioration in terms of metal or alloy thickness loss over time and helps distinguish between uniform corrosion and localised corrosion.

Corrosion Rate Calculation Formula
The most common way to determine corrosion rate is through the weight loss method, which measures the change in sample mass after exposure.
Corrosion Rate Formula (in mm/y):
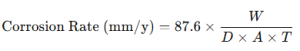
Where:
- W = weight loss (mg)
- D = density (g/cm³)
- A = exposed area (cm²)
- T = time of exposure (hours)
Alternatively, corrosion can be expressed in mils per year (mpy) using:
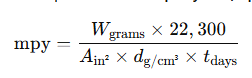
For short-term or field evaluations, the thickness loss method is used:
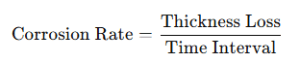
Units Used in Corrosion Rate Measurement
Corrosion rates are expressed in:
- mils per year (mpy) – commonly used in North America (1 mil = 0.001 inch)
- millimeters per year (mm/y) – standard SI unit
Factors Affecting Corrosion Rate
The corrosion rate is influenced by several environmental and material factors, including:
- Humidity and oxygen level affect oxidation reactions.
- Chloride concentration accelerates pitting in stainless steel.
- Temperature and pH influence corrosion kinetics.
- Alloy composition determines inherent corrosion resistance.
Importance of Corrosion Rate Calculation
Calculating the corrosion rate enables engineers to plan maintenance schedules, predict service life, and ensure the prevention of failures. Accurate monitoring also supports cost-efficient asset management, allowing timely intervention before severe damage or downtime occurs.
Methods for Determining Corrosion Rate
- Weight Loss Method: Based on mass change after exposure, ideal for uniform corrosion studies.
- Electrochemical Methods: These include linear polarisation resistance (LPR) and electrochemical impedance spectroscopy (EIS) to calculate rates based on polarisation resistance.
- Immersion Tests: Used to evaluate corrosion under controlled environmental conditions.
- Mathematical Models: Predict corrosion for complex systems (e.g., nuclear or chemical processing environments) based on temperature, acidity, and other factors.
